The Effect of Branching On Melting and Boiling Points
- Recall that alkanes are hydrocarbons with the general formula CnH(n+2)
- Isomers of hydrocarbons show regular trends with their melting and boiling points
- Generally, the linear alkane isomer in each series (e.g. n-hexane) will have the highest boiling point, and the boiling point decreases as branching increases.
- This is because the intermolecular forces in liquids are proportional to surface area (due to London dispersion forces), and branching decreases surface area.
- For melting points, increased symmetry results in higher melting points since the more regular an alkane “brick” is, the easier it is to stack.
- Relative to the branched isomers, the linear alkane isomer also tends to have a relatively high melting point since it is more symmetrical.
- However, with extreme branching can also come added symmetry, which can result in anomalously high melting points. One example is neopentane (2,2-dimethylpropane) which has a much higher melting point (-16°C) than that of pentane (-130°C).
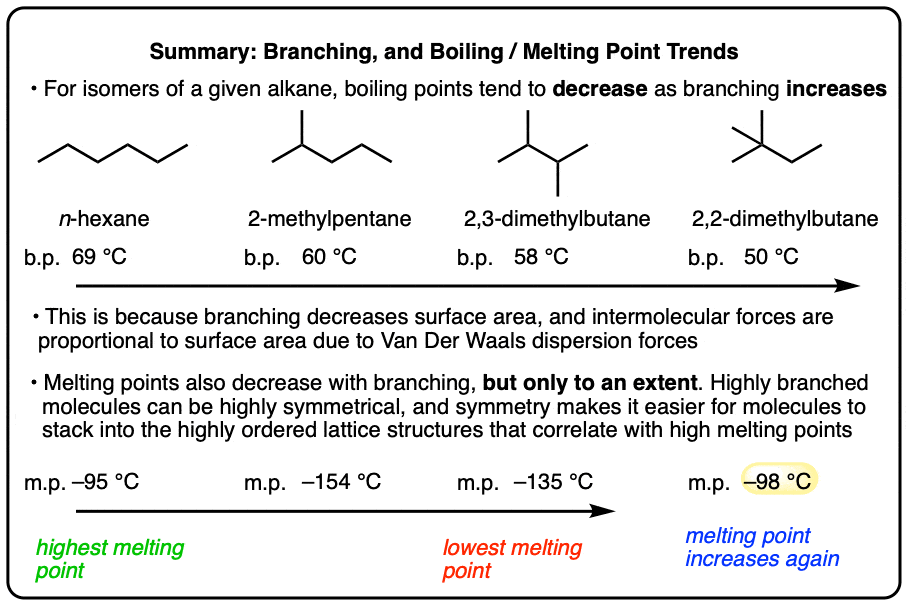
Table of Contents
-
- Better Stacking= Higher Melting Points
- Higher Surface Area = Higher Boiling Points
- The Key Trends
- Some More Experimental Data: Isomers of Heptane and Octane
- Notes
- Quiz Yourself!
1. Better Stacking = Higher Melting Points

The above photo shows what is perhaps one of the worst games of Tetris ever played. In my defence, the point wasn’t so much to play, but to take a photo of the great setup the Tech Model Railroad Club had set up next to the MIT museum, complete with a replica of the Green Building that you can play Tetris on. Truly the nerdiest place on earth.
Tetris is essentially a bricklaying game on a timer. You’re given tiles and you have to rotate them so that you make “lines” at the bottom, which promptly disappear when complete. If there are any spaces left in between, they remain – (for many examples, just look above). What makes Tetris hard is the arrangement of the different shapes. You often have to rotate them in order to get them to stack correctly.
If you want to make the game absurdly easy, just make it such that every tile looks like the one on the left. Or difficult, to the piece on the right.
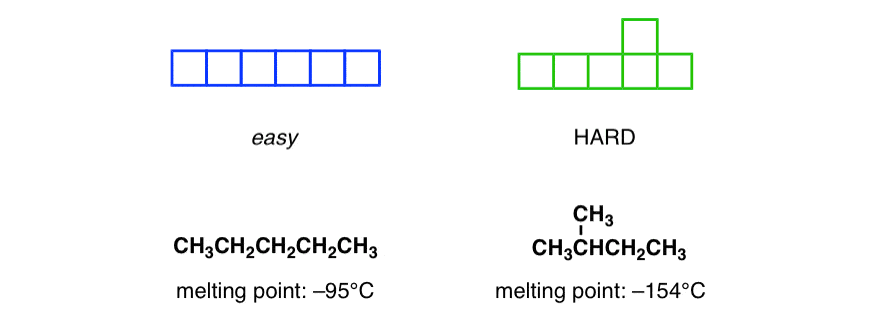
You’ll notice something – the simpler the pieces are, the easier they are to stack together, which provides a tighter fit with fewer spaces. Here, by putting a kink in the block, we make them harder to stack.
What’s this got to do with chemistry?
When compounds freeze, the process is a lot like stacking bricks. The more symmetrical the molecules are, the easier it will be and the fewer spaces there will be between the molecules. Fewer spaces = better stacking. Hence, when you compare hexane to its structural isomer, 2-methylpentane, hexane has a much higher melting point due to the regular arrangement of its structure.
Better stacking, higher melting point. Case closed. Right? Not quite.
2. Higher Surface Area = Higher Boiling Points
It’s a nice story: branching decreases melting point and boiling point. But it gets more complicated.
Look at these three examples of branched hexane derivatives (with hexane for comparison)
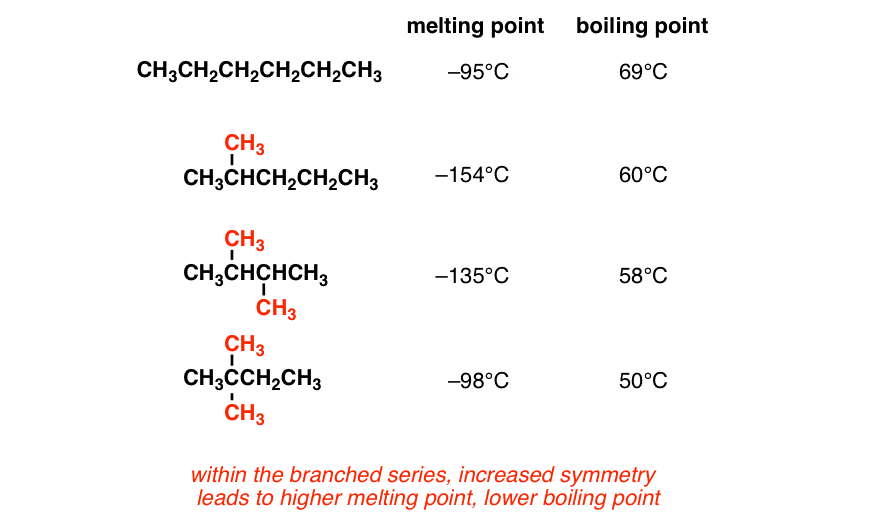
It looks like as we increase branching, we’re increasing melting point and decreasing boiling point. What’s going on?
Treat the n-hydrocarbon as a special case, and ignore it for the time being. Starting with the simplest branched compound, as you increase branching, you will increase the melting point, but decrease the boiling point. Why?
Going from “branched” to “highly branched” makes a molecule more compact and sphere-like. As the surface area of the molecule decreases (remember that spheres have the lowest surface area/volume ratio of any shape) they will become more compact and thus easier to pack. This explains the melting point phenomenon.
What about boiling point?
Boiling point is related to the forces between molecules, which in the case of hydrocarbons is Van Der Waals interactions. If you’ve ever seen microscope images of a gecko’s feet – which allow it to climb walls – you’ll see that there is no adhesive but the pads contain a tremendous amount of surface area. It’s all about the Van der Waals interactions.
As we decrease surface area, we are going to decrease intermolecular Van Der Waals interaction and therefore decrease boiling point.
3. The Key Relationships
So here are the relationships:
linear versus branched —> higher melting/boiling points due to better stacking and surface area contact.
highly branched vs. branched —> more sphere-like -> better stacking -> higher melting point
highly branched vs. branched —>more sphere-like – -> lower surface area —> lower boiling point.
If this all seems rather ambiguous, contradictory, and imprecise, well, you have a point. It’s not a straightforward topic. As a final example, I give you 2,2,3,3,-tetramethylbutane. An isomer of isooctane (gasoline) with melting point 95 °C. Further proof, as if more was needed, that predicting melting/boiling points from chemical structures can be a fool’s errand.
4. Some More Experimental Data: Isomers of Heptane and Octane
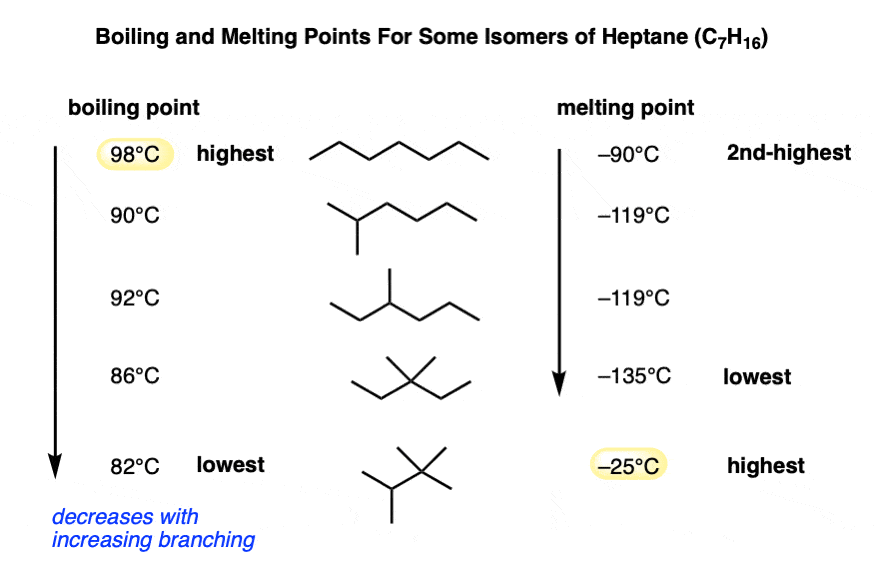
Looking at some experimental data, here are some isomers of heptane.
Note how the boiling point decreases as branching decreases (less surface area!) and the melting point also decreases with branching, but only to a point. The isomer 2,2,3-trimethylbutane has a higher melting point (-25°C) than linear heptane.
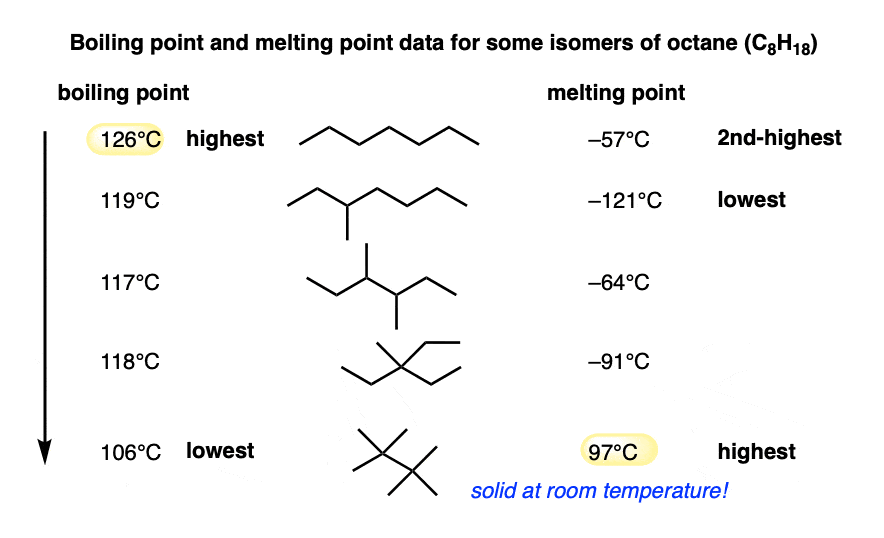
A similar phenomenon is observed for isomers of octane (C8H18). Note how the boiling points decrease with branching, but it’s a different story with the melting points. In particular, 2,2,3,3-tetramethylbutane is a solid at room temperature owing to its highly symmetrical structure.
Notes
Quiz Yourself!
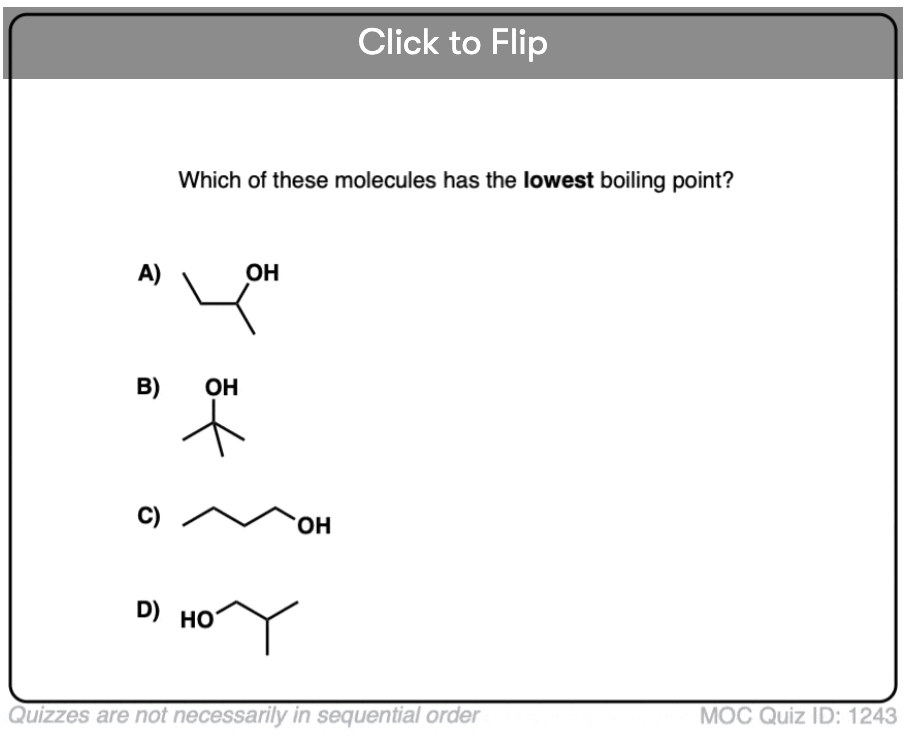
Become a MOC member to see the clickable quiz with answers on the back.
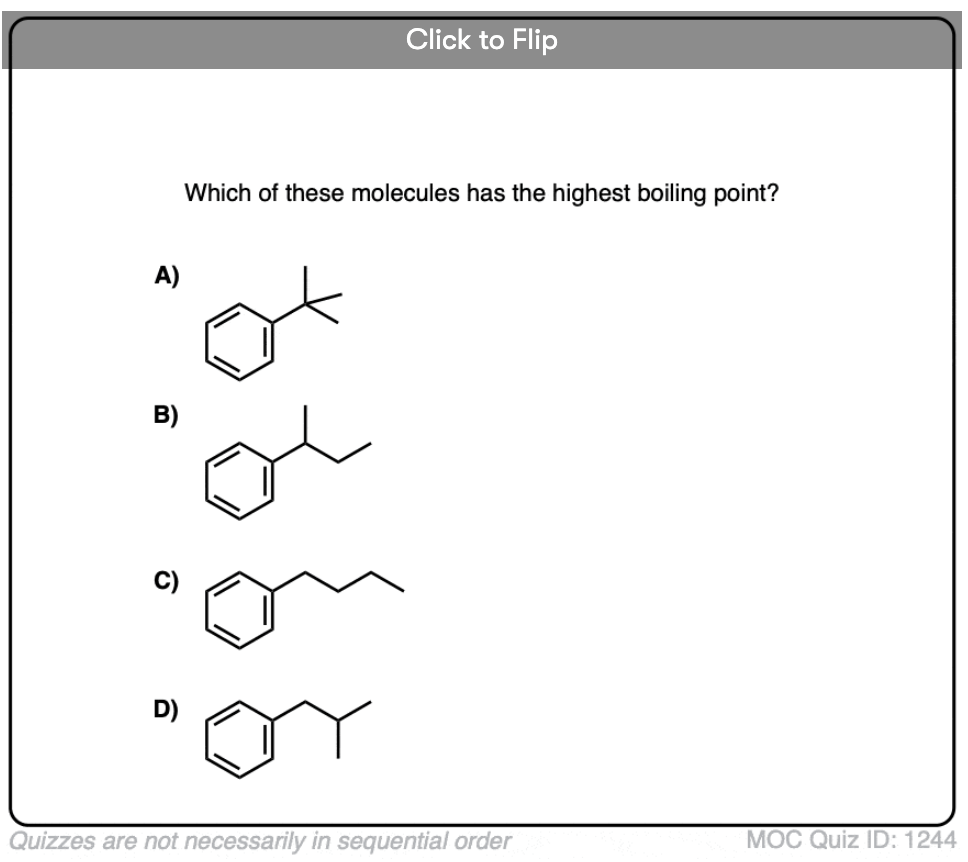
Become a MOC member to see the clickable quiz with answers on the back.
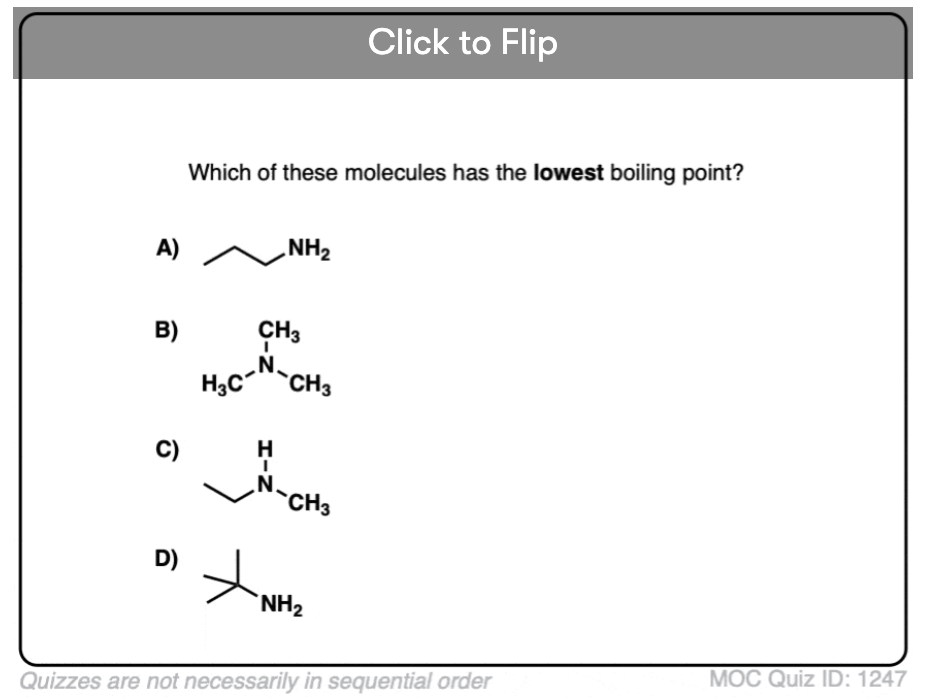
Become a MOC member to see the clickable quiz with answers on the back.
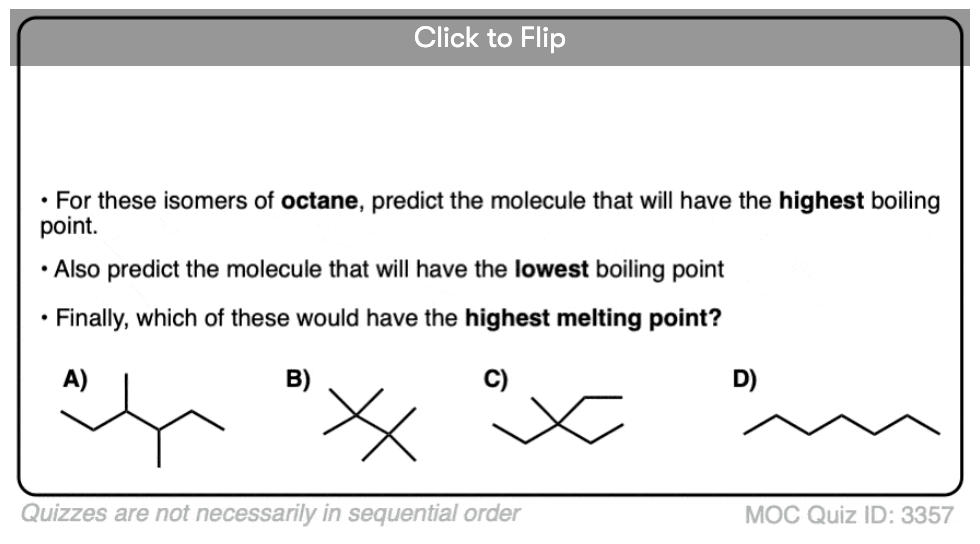
Become a MOC member to see the clickable quiz with answers on the back.
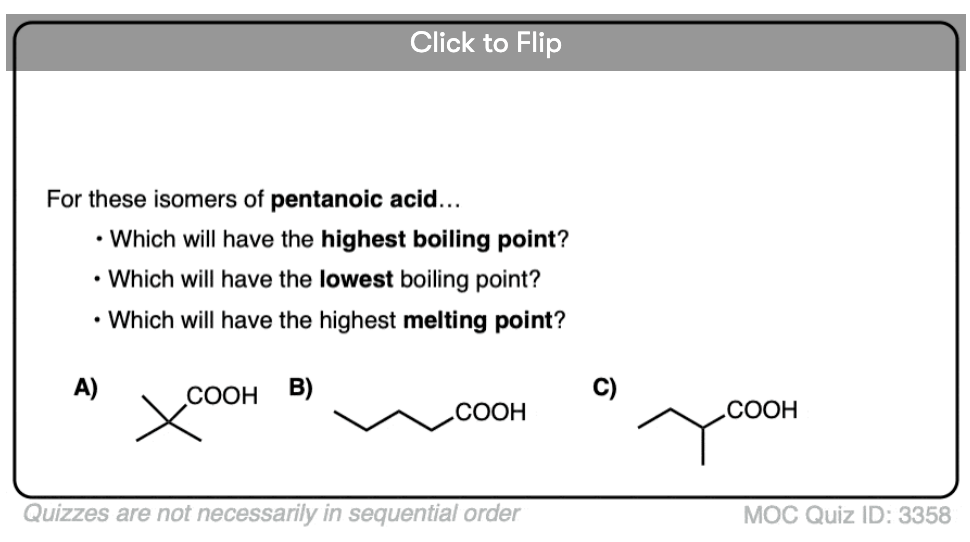
Become a MOC member to see the clickable quiz with answers on the back.
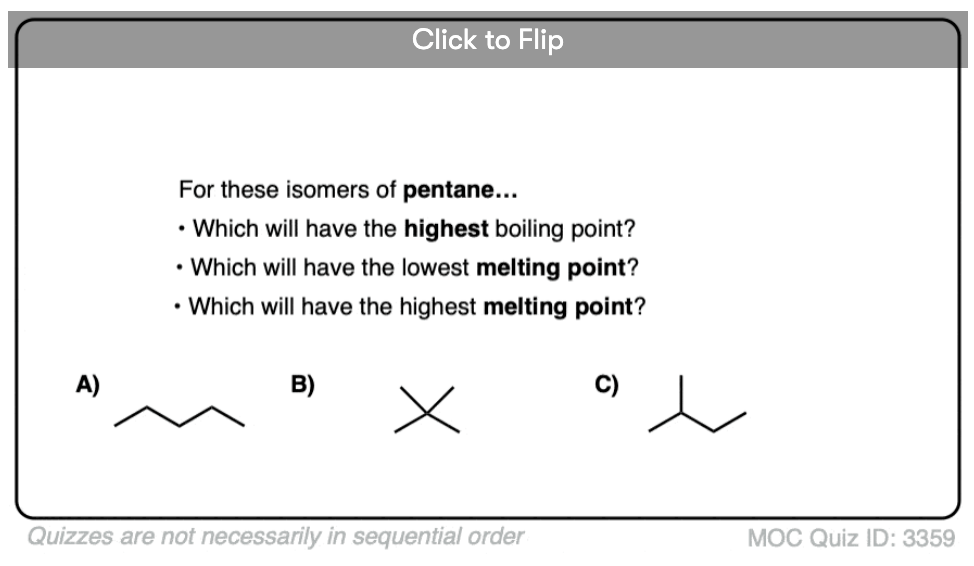
Become a MOC member to see the clickable quiz with answers on the back.